Tsung-yen Hsieh, Raj D. Dedhia, Whitney Chiao, Harley Dresner, Ruth J. Barta, Sofia Lyford-Pike, David Hamlar, Scott J. Stephan, Warren Schubert, and Peter A. Hilger
Published Online:15 May 2020: https://doi.org/10.1089/fpsam.2020.0185
Abstract
COVID-19 is an emerging viral illness that has rapidly transmitted throughout the world. Its impact on society and the health care system has compelled hospitals to quickly adapt and innovate as new information about the disease is uncovered. During this pandemic, essential medical and surgical services must be carried out while minimizing the risk of disease transmission to health care workers. There is an elevated risk of COVID-19 viral transmission to health care workers during surgical procedures of the head and neck due to potential aerosolization of viral particles from the oral cavity/naso-oropharynx mucosa. Thus, patients with facial fractures pose unique challenges to the variety of injuries and special considerations, including triaging injuries and protective measures against infection. The proximity to the oral cavity/naso-oropharyngeal mucosa, and potential for aerosolization of secretions containing viral particles during surgical procedures make most patients undergoing operative interventions for facial fractures high risk for COVID-19 transmission. Our proposed algorithm aims to balance patient care with patient/medical personnel protection as well as judicious health care utilization. It stratifies facial trauma procedures by urgency and assigns a recommended level of personal protective equipment, extreme or enhanced, incorporating current best practices and existing data on viral transmission. As this pandemic continues to evolve and more information is obtained, the protocol can be further refined and individualized to each institution.
Introduction
Coronavirus disease 2019 (COVID-19) is an insidious viral illness caused by the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) virus with a wide range of pulmonary manifestations that can vary from asymptomatic to lethal respiratory failure. With the first case reported in December 2019 and the disease declared a pandemic by the World Health Organization (WHO) 3 months later, it has now permeated our society and way of living.1,2
Health care teams have garnered attention as hospitals are reaching or exceeding their ability to care for afflicted patients. Personal protective equipment (PPE) supplies are being depleted, and increasingly, medical personnel are becoming patients themselves. An early study from China reported that health care workers with hospital-acquired COVID-19 constituted nearly one-third of their COVID-19 positive patients.1 In Italy and Spain, the two European countries most affected by COVID-19, health care workers are also vulnerable. In Spain alone, health care workers account for ∼14% of COVID-19 positive individuals.3,4
Otolaryngologists and other specialties that treat pathology of the head, neck, and airway are particularly vulnerable in light of the existing knowledge of COVID-19's mode of transmission. There is a high viral burden in the nose and the aerosolized form of the virus can persist for up to 3 h in the air and 48 to 72 h on select surfaces.5 Elective visits and surgeries are being cancelled in accordance with state, federal, and professional surgical associations' guidelines. However, certain circumstances such as facial trauma may preclude postponing intervention. We present a protocol to standardize facial trauma triage and precautions to impose to appropriately care for patients while minimizing risk to surgeons.
Protocol
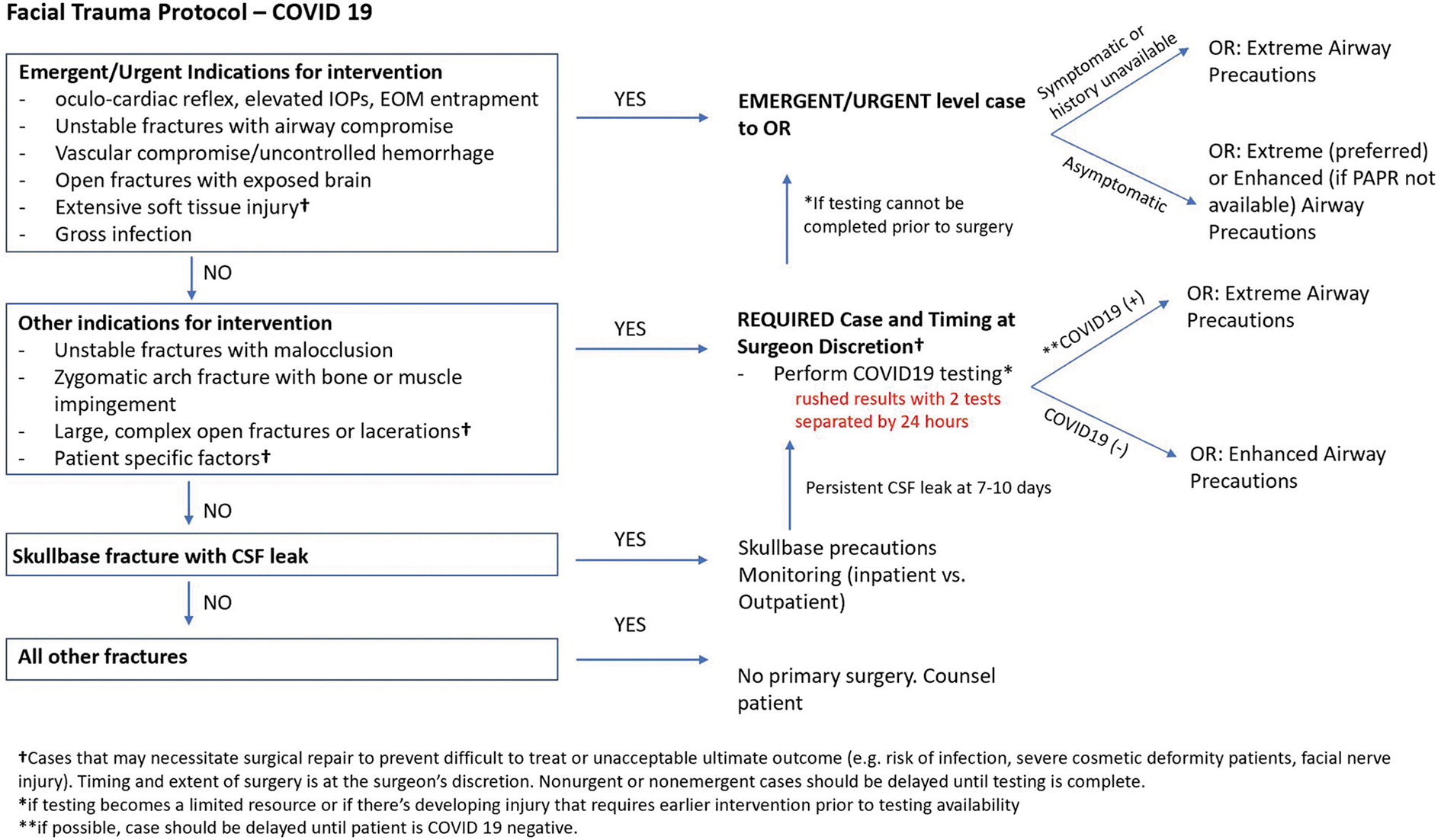
The goal of our proposed protocol (Fig. 1) is to balance patient care with patient/health care team protection as well as judicious health care utilization given finite resources. The management of facial fractures is particularly high risk given the viral load within the oral cavity/nasal–oropharyngeal mucosa, and the surgical instrumentation that is likely to aerosolize viral particles.
Fig. 1. Protocol for facial trauma triage and precautions during COVID-19 pandemic.
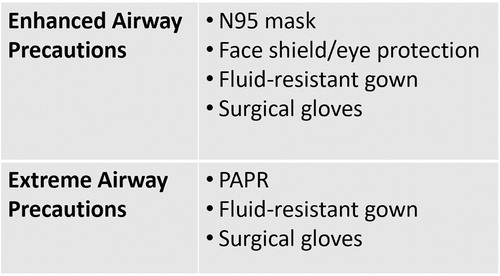
For our protocol, we utilized two sets of precautions: extreme versus enhanced airborne precautions (Fig. 2). Extreme airborne precautions include powered air-purifying respiratory (PAPR), fluid-resistant gown, and surgical gloves. Enhanced airborne precautions include N95 mask, face shield/eye protection, fluid-resistant gown, and surgical gloves.6
Fig. 2. Airway precautions designation.
For facial fractures wherein the morbidity and/or mortality is significant if no surgical intervention is provided urgently or emergently, there is usually not enough time for COVID-19 testing to be completed before proceeding to the operating room (OR). In cases wherein the patient is symptomatic or if the clinical history simply cannot be obtained, we recommend extreme airborne precautions. If the patient is asymptomatic, we prefer extreme airborne precautions if available, but enhanced airborne precautions can reasonably be substituted if PAPRs are limited. As an aside, facial soft tissue injuries are highly variable with a large spectrum of functional and cosmetic concerns depending on the structures involved. In these situations, the consideration for the need for surgical intervention, timing (emergent/urgent, vs. delayed surgery after COVID-19 testing), and extent of surgery should be determined on a case-by-case scenario.
Although some facial traumatic injuries are not emergent, they may necessitate surgical repair to prevent unacceptable sequelae that require more resources to treat than those required by the initial fracture. These sequelae can include infection, permanent functional deformity, and severe cosmetic deformity if left untreated. In these situations, surgery can often be performed in a less urgent manner and would allow time for COVID-19 testing to be completed. The turnaround time for COVID-19 testing is ∼24 h, although times may vary between laboratories and across health systems.7,8 Following guidelines from the Stanford University protocol, we recommend 48 h of preoperative testing that includes two COVID-19 tests 24 h apart. If both tests are negative, then surgery can proceed with enhanced airborne precautions. If one or both tests return positive, we recommend extreme airborne precautions.9 Considerations in surgical technique will be addressed in the Discussion section that follows.
In cases of skull base injury and cerebrospinal fluid (CSF) leakage, we recommend skull base precautions and monitoring. However, if the CSF leak persists after 7 days, the risk of meningitis increases.10,11 These patients should then proceed with surgery after preoperative COVID-19 testing is completed to determine the level of precautions needed.
For injuries that are nonemergent and do not cause significant unacceptable functional and/or cosmetic sequelae, the patient should be observed and/or operative intervention should be delayed. Patients should be counseled thoroughly on the risks and benefits of delaying operative intervention given the ongoing COVID-19 pandemic.
We acknowledge the inherent limitations of this protocol as PPE resources, caseload volumes, hospital capacity, and staffing availability are highly variable between institutions. Our objective is for this protocol to serve as a guide to triage and manage facial fractures in the current COVID-19 pandemic. Ultimately as this pandemic continues to evolve, this protocol can be further refined to provide a tailored approach to meet the specific demands of an institution.
Discussion
In mid-March 2020, the U.S. Surgeon General and the Centers for Medicare & Medicaid Services (CMS) recommended that elective and nonessential procedures, including surgeries, be postponed or cancelled in light of the COVID-19 outbreak. To date, more than two-thirds of U.S. states have released statements and executive orders in compliance with these recommendations.12
There is currently no consensus among professional surgical associations regarding the timing of elective surgeries. The American College of Surgeons (ACS) states that the timing of a nonemergent procedure is a nuanced decision that requires consideration of medical necessity, surgical expertise, feasibility, and available resources. It cites the Elective Surgery Acuity Scale as a framework to triage surgery, generally recommending against postponing high-acuity surgeries, whereas low- to intermediate-acuity surgeries may be postponed or performed at an ambulatory surgery center.13
Within otolaryngology, the American Academy of Otolaryngology-Head and Neck Surgery (AAO-HNS) has published more stringent measures given the high viral burden in the nasal cavity and nasopharynx. There is also mounting evidence from China, Italy, and Iran that otolaryngologists are at increased risk of becoming infected.5,14 The recommendation by AAO-HNS is that all procedures and examinations, whether in the office or the OR, should be limited to those that are “time-sensitive and emergent” with attention to the appropriate PPE and disinfection of equipment in the evaluation of and during all procedures for patients.14 This includes flexible laryngoscopy and nasal endoscopy. This position has since been reinforced by Arbeitsgemeinschaft für Osteosynthesefragen–Craniomaxillofacial (AO-CMF).15
The American Dental Association has released guidelines that define dental emergencies, including soft tissue infections or facial trauma that risk obstructing the airway, but does not specify management or protective measures.16 Vukkadala et al. published a comprehensive overview of otolaryngologic considerations, but did not address facial trauma. Recommendations from AO-CMF include preferred treatment and specific techniques to minimize risks for facial trauma by region, but does not explicitly describe how to triage or manage the timing for facial trauma operative intervention.15
Our protocol delineates the triage of patients and precautions to be taken in the setting of facial trauma to better allocate limited hospital resources while balancing long-term patient outcomes and the protection of medical personnel.
Current PPE recommendations
The most recent guidelines from the Stanford University School of Medicine advocate for caution as more information is being gathered. Emergent cases require PPE that may include PAPR.9 Patients requiring urgent surgery are recommended to have 48 h of preoperative testing that includes two COVID-19 tests 24 h apart. The turnaround time for COVID-19 results varies by state and by laboratory. State laboratories generally have results within 24 h, but may take up to 72 h; commercial laboratories are more variable, with results taking up to 4 or 5 days.7,8 The Stanford University protocol recommends PAPRs or a combination of an N95 mask with eye protection for all OR staff if a patient is found to be COVID-19 positive.6
AO-CMF recommends at minimum, an N95 mask with protective eyewear or PAPR in addition to gown, gloves, and a hat for all procedures and surgical interventions.15 The American Society of Anesthesiologists (ASA) also recognizes the increased risk of viral aerosolization and transmission from airway manipulation including endotracheal intubation. N95 masks, protective eyewear, gowns, and gloves are recommended. The ASA goes one step further and suggests conservation of PPE by prioritizing personnel at highest risk of COVID-19 exposure or professionals in the highest risk categories.17
In the setting of trauma, the ACS recommends preemptive PPE for droplet precautions for all patients. Patients with upper respiratory symptoms should don a face mask. They recognize the need for hospital policies for airway management in the trauma setting and for operative intervention in COVID-19 positive or COVID-19 status indeterminate patients, and recommend that hospitals create such policies should none exist.18
Amid the COVID-19 pandemic, medical personnel are confronted with the stark reality of the finite supply of PPE, including respirators. The Centers for Disease Control and Prevention (CDC) has developed an approach for conserving resources while reducing the exposure risk to the health care team. Initial measures include using respirators with an expired shelf life or those approved for occupational use in other countries. Notably, the CDC does not recommend the use of respirators past their shelf life in the surgical setting. When those options are no longer viable, the next options are limited reuse of N95 masks, use of respirators with an expired shelf life by National Institute for Occupational Safety and Health (NIOSH) standards, or prioritizing N95 masks and PAPRs for those with the highest potential exposure. This includes aerosol-generating procedures.19
We recognize that with increasing demand and finite resources, the availability of PAPRs will likely become significantly limited. The AO-CMF guidelines have reported alternative strategies in these situations such as the Stryker (Kalamazoo, MI) Flute system with an FFP3 mask, or FF2/FF3 mask combined with goggles and a hood.15
Finally, in the event that respirators are no longer available, the CDC proposes designating medical personnel who have recovered from COVID-19 to provide care, while restricting those at increased risk of a severe illness from COVID-19. As a final measure, medical personnel may have to wear non-NIOSH approved or homemade masks, although the CDC no longer formally recommends this as of April 2020.19
Airway and anesthesia considerations
After the release of CMS guidelines, AAO-HNS released its best practices and recommendations for transnasal and transoral examinations. Topical medications should be administered through pledgets rather than spray, which risk aerosolizing the virus.20
Once the patient has proceeded to the OR, permitted staff should ideally wait outside the operating suite for 20 to 30 min after intubation with aerosol generation. This is the time required to remove airborne contaminants with 99% and 99.9% efficiency, respectively; however, in reality the wait time will be determined by the urgency of the case and the risk incurred by the surgery itself.21
AAO-HNS and the British Association of Otorhinolaryngologists–Head and Neck Surgeons and British Academic Conference in Otolaryngology (ENT UK) have released official statements regarding the timing and management of tracheostomies. AAO-HNS is more explicit in stating that tracheostomies should only be performed in patients with a stable ventilatory status, preferably with known COVID-19 negative status, and should be performed no sooner than 2 to 3 weeks after intubation.22 Open versus percutaneous tracheostomy may be hospital and surgeon specific, but open tracheostomy may be preferred due to a theoretical decreased risk of aerosolization of secretions.15,23 However, these considerations may need to be adjusted depending on the urgency of trauma cases and the ability to secure an airway in these situations.
Both AAO-HNS and ENT UK highlight the need for greater attention in post-tracheostomy cares to minimize droplet transmission. This includes keeping the tracheostomy tube cuff appropriately inflated, favoring in-line suctioning, maintaining a closed circuit, and deferring routine post-tracheostomy changes until COVID-19 status is negative.22,23
Surgical considerations
The AO-CMF has provided detailed guidelines regarding the management of facial fractures and some considerations to reduce risk of COVID-19 transmission (Table 1).15
Several general considerations in the decision for surgery can be stated at this time. Procedures should be limited to facial fractures in which a delay in management could affect ultimate outcome (Fig. 1). Surgeon-specific factors include age >60 years, immunosuppression, cardiac comorbidities, chronic pulmonary disorder, or multiple other medical comorbidities. In these instances, surgeons should avoid performing repairs of facial traumatic injuries when possible. The number of residents and ancillary staff in the OR should be limited whenever possible. Proper PPE and training for all members of the operative team are required. Lastly, surgical scrubs worn during the procedure should be changed immediately afterward. Existing evidence suggests that if infected, surgeons are at increased risk of a more severe illness, intensive care unit admission, and increased mortality.1,24,25
One special consideration worth exploring is extending these protections to reduce exposure in the outpatient setting. Access to PAPRs and N95 masks is likely to be limited in outpatient clinics. Long-term maxillomandibular fixation (MMF) should, therefore, be avoided if possible, to avoid transmission during in-office MMF adjustments and removal.
Lastly, there has been evidence of virucidal properties of chlorhexidine gluconate and povidone-iodine mouthwash against respiratory and oral tract pathogens.26–29 Although there is no current evidence for their effects on the COVID-19 patient population, preoperative swish and spit with these agents may be considered to potentially decrease viral load in the oral cavity/oropharynx.
Conclusions
COVID-19, caused by SARS-CoV-2, is an emerging pathogen with a global impact creating new health care challenges. Special considerations are needed in the triage and precautions for management of facial trauma given the high viral burden of the oral cavity/naso-oropharyngeal mucosa and the risk of aerosolization of viral particles with instrumentation. We have created a protocol to triage facial trauma and designate appropriate PPE according to risk per procedure; this takes into consideration hospital resource allocation while balancing long-term patient outcomes and medical personnel protection. As the COVID-19 pandemic continues to evolve, we hope this protocol can be further refined and individualized to each institution.
Author Disclosure Statement
No competing financial interests exist.
Funding Information
No funding was received for this article.
The AO-CMF has provided detailed guidelines regarding the management of facial fractures and some considerations to reduce risk of COVID-19 transmission (Table 1).15
References
- 1. Wang D, Hu B, Hu C, et al. Clinical Characteristics of 138 Hospitalized Patients With 2019 Novel Coronavirus-Infected Pneumonia in Wuhan, China. JAMA. 2020;323(11):1061–1069. Crossref, Medline, Google Scholar
- 2. World Health Organization. WHO Director-General's opening remarks at the media briefing on COVID-19. World Health Organization. 2020. https://www.who.int/dg/speeches/detail/who-director-general-s-opening-remarks-at-the-media-briefing-on-covid-19 Accessed March 30, 2020. Google Scholar
- 3. Editorial Staff. Elenco dei Medici caduti nel corso dell'epidemia di Covid-19 [List of doctors who died during the Covid-19 epidemic]. Federazione Nazionale degli Ordini dei Medici Chirurghi e degli Odontoiatri. 2020. https://portale.fnomceo.it/elenco-dei-medici-caduti-nel-corso-dellepidemia-di-covid-19/ Accessed March 30, 2020. Google Scholar
- 4. Minder R, Peltier E. Virus knocks thousands of health workers out of action in Europe. The New York Times. 2020. https://www.nytimes.com/2020/03/24/world/europe/coronavirus-europe-covid-19.html Accessed March 30, 2020. Google Scholar
- 5. Zou L, Ruan F, Huang M, et al. SARS-CoV-2 viral load in upper respiratory specimens of infected patients. N Engl J Med 2020;382(12):1177–1179. Crossref, Medline, Google Scholar
- 6. Vukkadala N, Qian ZJ, Holsinger FC, Patel ZM, Rosenthal E. COVID-19 and the otolaryngologist—preliminary evidence-based review. Laryngoscope. [Epub ahead of print]; DOI: 10.1002/lary.28672. Google Scholar
- 7. LabCorp. LabCorp's Testing For COVID-19. LabCorp. 2020. https://www.labcorp.com/assets-media/2330 Accessed March 30, 2020. Google Scholar
- 8. Kaplan A, Georgiev P, Enright M, Oakes C. What to do if you are concerned you have COVID-19, according to state health departments. NBC News. 2020. https://www.nbcnews.com/health/health-news/coronavirus-testing-information-covid-19-tests-according-state-health-departments-n1158041 Accessed March 30, 2020. Google Scholar
- 9. ENTtoday. Otolaryngologists may contract Covid-19 during surgery. ENTtoday. 2020. https://www.enttoday.org/article/otolaryngologists-may-contract-covid-19-during-surgery/ Accessed March 24, 2020. Google Scholar
- 10. Leech PJ, Paterson A. Conservative and operative management for cerebrospinal-fluid leakage after closed head injury. Lancet. 1973;1(7811):1013–1016. Crossref, Medline, Google Scholar
- 11. Mincy JE. Posttraumatic cerebrospinal fluid fistula of the frontal fossa. J Trauma. 1966;6(5):618–622. Crossref, Medline, Google Scholar
- 12. Ambulatory Surgery Center Association. State Guidance on Elective Surgeries. Ambulatory Surgery Center Association. 2020. https://www.ascassociation.org/asca/resourcecenter/latestnewsresourcecenter/covid-19/covid-19-state Accessed April 6, 2020. Google Scholar
- 13. American College of Surgeons. COVID-19: Guidance for triage of non-emergent surgical procedures. American College of Surgeons. 2020. https://www.facs.org/covid-19/clinical-guidance/triage Accessed March 24, 2020. Google Scholar
- 14. American Academy of Otolaryngology—Head and Neck Surgery. Otolaryngologists and the COVID-19 Pandemic. American Academy of Otolaryngology—Head and Neck Surgery. 2020. https://www.entnet.org/content/otolaryngologists-and-covid-19-pandemic Accessed March 24, 2020. Google Scholar
- 15. Grant M, Schramm A, Strong B, et al. AO CMF international task force recommendations on best practices for maxillofacial procedures during COVID-19 pandemic. 2020. Available at https://aocmf3.aofoundation.org/-/media/project/aocmf/aocmf/files/covid-19/ao_cmf_covid-19_task_force_guidelines.pdf?la=en&hash=C2B89E1E6E9AB72EBF386C747D3BC74CF1009C1E Accessed March 30, 2020. Google Scholar
- 16. American Dental Association. What constitutes a dental emergency? American Dental Association. 2020. https://success.ada.org/~/media/CPS/Files/Open%20Files/ADA_COVID19_Dental_Emergency_DDS.pdf?_ga=2.8349874.1806649990.1586205522-975892202.1586205522 Accessed March 30, 2020. Google Scholar
- 17. American Society of Anesthesiologists. The use of personal protective equipment by anesthesia professionals during the COVID-19 pandemic. American Society of Anesthesiologists. 2020. https://www.asahq.org/about-asa/newsroom/news-releases/2020/03/the-use-of-personal-protective-equipment-by-anesthesia-professionals-during-the-covid-19-pandemic Accessed March 30, 2020. Google Scholar
- 18. American College of Surgeons Committee on Trauma. Maintaining Trauma Center Access & Care during the COVID-19 Pandemic: Guidance Document for Trauma Medical Directors. American College of Surgeons. 2020. https://www.facs.org/quality-programs/trauma/maintaining-access Accessed March 30, 2020. Google Scholar
- 19. Centers for Disease Control and Prevention. Strategies for optimizing the supply of N95 respirators. Centers for Disease Control and Prevention. 2020. https://www.cdc.gov/coronavirus/2019-ncov/hcp/respirators-strategy/crisis-alternate-strategies.html Accessed March 30, 2020. Google Scholar
- 20. American Academy of Otolaryngology—Head and Neck Surgery. Academy Supports CMS, Offers Specific Nasal Policy. American Academy of Otolaryngology—Head and Neck Surgery. 2020. https://www.entnet.org/content/academy-supports-cms-offers-specific-nasal-policy Accessed March 24, 2020. Google Scholar
- 21. Centers for Disease Control and Prevention. Appendix B. Air guidelines for environmental infection control in health-care facilities. Centers for Disease Control and Prevention. 2003. https://www.cdc.gov/infectioncontrol/guidelines/environmental/appendix/air.html Accessed March 30, 2020. Google Scholar
- 22. Airway and Swallowing Committee of the American Academy of Otolaryngology-Head and Neck Surgery. Tracheotomy recommendations during the COVID-19 pandemic. American Academy of Otolaryngology—Head and Neck Surgery. 2020. https://www.entnet.org/content/tracheotomy-recommendations-during-covid-19-pandemic Accessed March 30, 2020. Google Scholar
- 23. ENT UK. Guidance for surgical tracheostomy and tracheostomy tube change during the COVID-19 pandemic. ENT UK. 2020. https://www.entuk.org/tracheostomy-guidance-during-covid-19-pandemic Accessed March 30, 2020. Google Scholar
- 24. Zhou F, Yu T, Du R, et al. Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: a retrospective cohort study. Lancet. 2020;395(10229):1054–1062. Crossref, Medline, Google Scholar
- 25. Wu C, Chen X, Cai Y, et al. Risk factors associated with acute respiratory distress syndrome and death in patients with coronavirus disease 2019 pneumonia in Wuhan, China. JAMA Intern Med. [Epub ahead of print]; DOI:10.1001/jamainternmed.2020.0994. Google Scholar
- 26. Bernstein D, Schiff G, Echler G, Prince A, Feller M, Briner W. In vitro virucidal effectiveness of a 0.12%-chlorhexidine gluconate mouthrinse. J Dent Res. 1990;69(3):874–876. Crossref, Medline, Google Scholar
- 27. Eggers M, Eickmann M, Zorn J. Rapid and effective virucidal activity of povidone-iodine products against middle east respiratory syndrome coronavirus (MERS-CoV) and modified vaccinia virus ankara (MVA). Infect Dis Ther. 2015;4(4):491–501. Crossref, Medline, Google Scholar
- 28. Lim KS, Kam PC. Chlorhexidine—pharmacology and clinical applications. Anaesth Intensive Care. 2008;36(4):502–512. Crossref, Medline, Google Scholar
- 29. Eggers M, Koburger-janssen T, Eickmann M, Zorn J. In vitro bactericidal and virucidal efficacy of povidone-iodine gargle/mouthwash against respiratory and oral tract pathogens. Infect Dis Ther. 2018;7(2):249–259. Crossref, Medline, Google Scholar